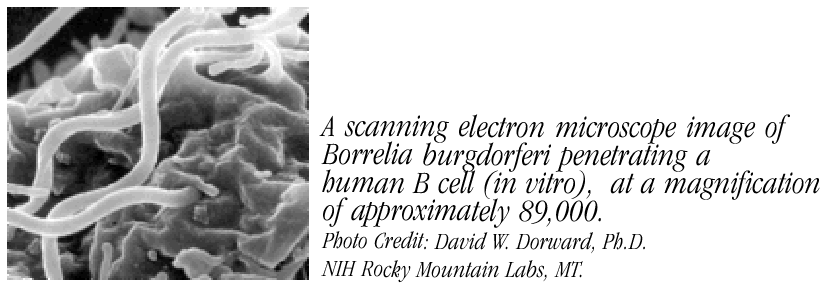
 We found that B. burgdorferi actively attaches to, invades, and kills human B and T lymphocytes.
We found that B. burgdorferi actively attaches to, invades, and kills human B and T lymphocytes.
The Lyme Times 2000 Spring;28:86-88 (in cache)
Lyme Disease Resource Center
P.O.Box 707
Weaverville, CA 96093 - 0707
USA
Likewise in PCR reaction for detection of Bb DNA in skin lesions of
early and late Lyme borreliosis von Stedingk
in Eur J Clin Microbiol Infec Dis 1995 Jan ; 14 ;1-5 found that over 60%
of these skin biopsies had Bb DNA and after antibiotic treatment were negative
but staining could not demonstrate Bb. Where were they?
Infect Immun 1995 Sep;63(9):3491-6
Related
Articles, Books,
LinkOut
Binding of human plasminogen to Borrelia burgdorferi.
Hu LT, Perides G, Noring R, Klempner MS
Division of Geographic Medicine and Infectious Diseases, Tufts University School of Medicine, New England Medical Center, Boston, Massachusetts 02111, USA.
We studied the binding of plasminogen to Borrelia burgdorferi, a spirochete
which causes Lyme disease and produces no endogenous proteases which digest
extracellular matrix proteins.
PMID: 7642282, UI: 95369905
J Immunol 1996 Oct 1;157(7):2998-3005
Related
Articles,Books,
LinkOut
Human dendritic cells phagocytose and process Borrelia burgdorferi.
Filgueira L, Nestle FO, Rittig M, Joller HI, Groscurth P
Department of Anatomy, University of Zurich, Switzerland.
There is strong evidence that the immune response to Borrelia burgdorferi (Bb) contributes to the pathogenesis of Lyme disease. Bb are transmitted by ticks to the skin, which is particularly rich in dendritic cells (DC). The initial reaction of these APCs may already set the course to immune pathogenesis.
To study the role of dendritic cells,
PMID: 8816408, UI: 96413246
Infect Immun 1991 Feb;59(2):671-8
Related
Articles
The later stages of infection by the Lyme disease pathogen, Borrelia burgdorferi, are characterized by the persistence of the organism in individuals possessing a strong anti-Borrelia immune response. This suggests that the organism is sequestered in a tissue protected from the immune system of the host or there is a reservoir of the organism residing within the cells of the host. In this report, the ability of B. burgdorferi to gain entrance into human umbilical vein endothelial cells was explored as a model for invasion. Incubation of B. burgdorferi with human umbilical vein endothelial cells at ratios ranging from 200:1 to 5,000:1 resulted in the intracellular localization of 10 to 25% of B. burgdorferi in 24 h. The intracellular location of the spirochetes was demonstrated by the incorporation of radiolabeled B. burgdorferi into a trypsin-resistant compartment and was confirmed by double-immunofluorescence staining which differentiated intracellular from extracellular organisms. Actin-containing microfilaments were required for the intracellular localization, indicating that the host cell participates in the internalization process. Activation of endothelial cells by agents known to increase the expression of several adhesion molecules had no effect on the interaction of B. burgdorferi with the endothelial monolayer. This indicates that the endothelial receptor for B. burgdorferi is constitutively expressed and that internalization is not dependent upon adhesion molecules whose expression is induced by inflammatory mediators. The demonstration of B. burgdorferi within endothelial cells suggest that intracellular localization may be a potential mechanism by which the organism escapes from the immune response of the host and may contribute to persistence of the organism during the later stages of Lyme disease.
PMID: 1987083, UI: 91100043
Lyme disease is a persistent low-density spirochetosis caused by Borrelia burgdorferi sensu lato. Although spirochetes causing Lyme disease are highly immunogenic in experimental models, the onset of specific antibody responses to infection is often delayed or undetectable in some patients. The properties and mechanisms mediating such immune avoidance remain obscure. To examine the nature and consequences of interactions between Lyme disease spirochetes and immune effector cells, we coincubated B. burgdorferi with primary and cultured human leukocytes.
 We found that B. burgdorferi actively attaches to, invades, and kills human B and T lymphocytes.
We found that B. burgdorferi actively attaches to, invades, and kills human B and T lymphocytes.
PMID: 9233657, UI: 97376886
Ann Rheum Dis 1998 Feb;57(2):118-21
Related
Articles, Books,
LinkOut
Detection of Borrelia burgdorferi by polymerase chain reaction in synovial membrane, but not in synovial fluid from patients with persisting Lyme arthritis after antibiotic therapy.
Priem S, Burmester GR, Kamradt T, Wolbart K, Rittig MG, Krause A
Charite University Hospital, Department of Medicine III, Rheumatology and Clinical Immunology, Berlin, Germany.
OBJECTIVES: To identify possible sites of bacterial persistence in patients with treatment resistant Lyme arthritis. It was determined whether Borrelia burgdorferi DNA may be detectable by polymerase chain reaction (PCR) in synovial membrane (SM) when PCR results from synovial fluid (SF) had become negative after antibiotic therapy. METHODS: Paired SF and SM specimens and urine samples from four patients with ongoing or recurring Lyme arthritis despite previous antibiotic therapy were investigated. A PCR for the detection of B burgdorferi DNA was carried out using primer sets specific for the ospA gene and a p66 gene of B burgdorferi. RESULTS: In all four cases, PCR with either primer set was negative in SF and urine, but was positive with at least one primer pair in the SM specimens. In all patients arthritis completely resolved after additional antibiotic treatment. CONCLUSIONS: These data suggest that in patients with treatment resistant Lyme arthritis negative PCR results in SF after antibiotic therapy do not rule out the intraarticular persistence of B burgdorferi DNA. Therefore, in these patients both SF and SM should be analysed for borrelial DNA by PCR as positive results in SM are strongly suggestive of ongoing infection.
PMID: 9613343, UI: 98276291
Hum Pathol 1996 Oct;27(10):1025-34
Related
Articles, Books,
LinkOut
Ultrastructural demonstration of spirochetal antigens in synovial fluid and synovial membrane in chronic Lyme disease: possible factors contributing to persistence of organisms.
Nanagara R, Duray PH, Schumacher HR Jr
Allergy-Immunology-Rheumatology Division, Department of Medicine, Faculty of Medicine, KhonKaen University, Thailand.
To perform the first systematic electronmicroscopic (EM) and immunoelectron microscopy (IEM) study of the pathological changes and the evidence of spirochete presence in synovial membranes and synovial fluid (SF) cells of patients with chronic Lyme arthritis. EM examination was performed on four synovial membrane and eight SF cell samples from eight patients with chronic Lyme disease. Spirochetal antigens in the samples were sought by IEM using monoclonal antibody to Borrelia burgdorferi outer surface protein A (OspA) as the immunoprobe. Prominent ultrastructural findings were surface fibrin-like material, thickened synovial lining cell layer and signs of vascular injury. Borrelia-like structures were identified in all four synovial membranes and in two of eight SF cell samples. The presence of spirochetal antigens was confirmed by IEM in all four samples studied (one synovial membrane and three SF cell samples). OspA labelling was in perivascular areas, deep synovial stroma among collagen bundles, and in vacuoles of fibroblasts in synovial membranes; and in cytophagosomes of mononuclear cells in SF cell samples. Electron microscopy adds further evidence for persistence of spirochetal antigens in the joint in chronic Lyme disease. Locations of spirochetes or spirochetal antigens both intracellulary and extracellulary in deep synovial connective tissue as reported here suggest sites at which spirochaetes may elude host immune response and antibiotic treatment.
PMID: 8892586, UI: 97047745
Interaction of Borrelia burgdorferi with peripheral blood fibrocytes, antigen-presenting cells with the potential for connective tissue targeting.
Grab DJ, Lanners H, Martin LN, Chesney J, Cai C, Adkisson HD, Bucala R
Tulane Regional Primate Research Center, Covington, Louisiana, USA. grab@cc.saga-u.ac.jp
BACKGROUND: Borrelia Burgdorferi has a predilection for collagenous tissue and can interact with fibronectin and cellular collagens. While the molecular mechanisms of how B. burgdorferi targets connective tissues and causes arthritis are not understood, the spirochetes can bind to a number of different cell types, including fibroblasts. A novel circulating fibroblast-like cell called the peripheral blood fibrocyte has recently been described. Fibrocytes express collagen types I and III as well as fibronectin. Besides playing a role in wound healing, fibrocytes have the potential to target to connective tissue and the functional capacity to recruit, activate, and present antigen to CD4(+) T cells. MATERIALS AND METHODS: Rhesus monkey fibrocytes were isolated and characterized by flow cytometry. B. burgdorferi were incubated with human or monkey fibrocyte cultures in vitro and the cellular interactions analyzed by light and electron microscopy. The two strains of B. burgdorferi studied included JD1, which is highly pathogenic for monkeys, and M297, which lacks the cell surface OspA and OspB proteins. RESULTS: In this study, we demonstrate that B. burgdorferi binds to both human and monkey (rhesus) fibrocytes in vitro. This process does not require OspA or OspB. In addition, the spirochetes are not phagocytosed but are taken into deep recesses of the cell membrane, a process that may protect them from the immune system. CONCLUSIONS: This interaction between B. burgdorferi and peripheral blood fibrocytes provides a potential explanation for the targeting of spirochetes to joint connective tissue and may contribute to the inflammatory process in Lyme arthritis.
PMID: 10072447, UI: 99172272
J Infect Dis 1992 Aug;166(2):440-4
Related
Articles
The Lyme disease spirochete, Borrelia burgdorferi, can be recovered long after initial infection, even from antibiotic-treated patients, indicating that it resists eradication by host defense mechanisms and antibiotics. Since B. burgdorferi first infects skin, the possible protective effect of skin fibroblasts from an antibiotic commonly used to treat Lyme disease, ceftriaxone, was examined. Human foreskin fibroblasts protected B. burgdorferi from the lethal action of a 2-day exposure to ceftriaxone at 1 microgram/mL, 10-20 x MBC. In the absence of fibroblasts, organisms did not survive. Spirochetes were not protected from ceftriaxone by glutaraldehyde-fixed fibroblasts or fibroblast lysate, suggesting that a living cell was required. The ability of the organism to survive in the presence of fibroblasts was not related to its infectivity. Fibroblasts protected B. burgdorferi for at least 14 days of exposure to ceftriaxone. Mouse keratinocytes, HEp-2 cells, and Vero cells but not Caco-2 cells showed the same protective effect. Thus, several eukaryotic cell types provide the Lyme disease spirochete with a protective environment contributing to its long-term survival.
PMID: 1634816, UI: 92340959
J Clin Microbiol 1994 Mar;32(3):715-20
Related
Articles, Books,
LinkOut
Isolation of Borrelia burgdorferi from biopsy specimens
taken from healthy-looking skin of patients with Lyme borreliosis.
Kuiper H, van Dam AP, Spanjaard L, de Jongh BM, Widjojokusumo A, Ramselaar TC, Cairo I, Vos K, Dankert J
Department of Medical Microbiology, Academic Medical Centre, University Hospital, University of Amsterdam, The Netherlands.
Erythematous skin lesions due to infection with Borrelia burgdorferi will often disappear without antibiotic treatment. The aim of the study was to assess whether after disappearance of the erythematous skin lesion B. burgdorferi is still present in the healthy-looking skin of untreated patients. In six patients, a skin biopsy specimen was taken at the site of a previous erythematous skin lesion 1 to 6 months after disappearance of the lesion. Four of them presented with early disseminated Lyme borreliosis. In one additional patient with early disseminated Lyme borreliosis, the site of a previous tick bite was biopsied. None of these patients had been treated with antibiotics before presentation. The cultures of the skin biopsy specimens of the seven patients showed growth of Borrelia species. By rRNA gene restriction analysis and genospecies-specific PCR, six isolates were classified as Borrelia garinii and one as Borrelia group VS461. These results show that B. burgdorferi can still be cultured from the skin after disappearance of the erythematous skin lesion or at the site of a previous tick bite.
PMID: 8195384, UI: 94253337
Eur J Clin Microbiol Infect Dis 1995 Jan;14(1):1-5
Related
Articles, Books,
LinkOut
Polymerase chain reaction for detection of Borrelia
burgdorferi DNA in skin lesions of early and late Lyme borreliosis.
von Stedingk LV, Olsson I, Hanson HS, Asbrink E, Hovmark A
Department of Clinical Microbiology, Karolinska Hospital, Stockholm, Sweden.
The aim of this study was to evaluate the polymerase chain reaction (PCR) as a diagnostic tool for Lyme borreliosis on large numbers of samples from clinically well-defined cases of early and late cutaneous borreliosis. Skin biopsy specimens from patients with erythema migrans and acrodermatitis chronica atrophicans were analysed blindly together with an equal number of control biopsies. Using two different dilutions of each DNA specimen increased the number of total positives detected. All of the 76 control biopsies were PCR negative. Biopsy specimens from 18 of 26 (69%) erythema migrans lesions and from 22 of 36 (61%) acrodermatitis chronica atrophicans lesions were PCR positive. Fourteen post-therapy biopsies from patients with acrodermatitis chronica atrophicans were all negative, supporting the opinion that antibiotic therapy is successful in this chronic manifestation of Lyme borreliosis.
PMID: 7729446, UI: 95246765
Zentralbl Bakteriol 1994 Jan;280(3):348-59
Related
Articles, Books,
LinkOut
Electron microscopy of Langerhans cells and Borrelia burgdorferi in Lyme disease patients.
Hulinska D, Bartak P, Hercogova J, Hancil J, Basta J, Schramlova J
Institute of Public Health, Prague, Czech Republic.
To investigate dermal and epidermal involvement in the presence of Borrelia burgdorferi and to analyze the role of Langerhans cells and keratinocytes, 14 cases of erythema chronicum migrans and two controls were studied by means of electron microscopy, using negative staining and sectioning techniques. Using immunoelectron microscopy and histochemistry, positive results for B. burgdorferi were disclosed in 5 cases of erythema chronicum migrans and 3 cases of neuroborreliosis which were confirmed by cultivation. We cultured 4 stains of B. burgdorferi from the skin, 1 from blood and 2 from cerebrospinal fluid in BSK medium. Near to the centre of erythema chronicum migrans with focal necrosis were both a dissolved basal membrane and keratinocyte desmosomes surrounding damaged B. burgdorferi cells in the epidermis. Markedly oedematous keratinocytes and Langerhans cells with B. burgdorferi were released into lymphocyte infiltrates. At the periphery of all erythema chronicum migrans lesions, keratinocytes were well preserved while all dendritic cells seemed to be vacuolated. Above foci of B. burgdorferi located perivascular or among collagen fibers, Langerhans cells were frequent and more granulated. The possible role of Langerhans cells in the identification and elimination of B. burgdorferi is discussed.
PMID: 8167429, UI: 94220807
Electron microscopy was used to present morphological events, which accompany the uptake of Borrelia garinii by polymorphonuclear leukocytes (PMN) and intracellular events of phagocytosis throughout 2 hours of contact. Phagocytosis of borreliae proceeded very quickly, and seemed to be independent of opsonins. Opsonized borreliae emitted membrane-bound blebs, which were attached and engulfed by PMN. Evidence of coiling and conventional phagocytosis were reported in the same cell. Coiling phagocytosis was time dependent (number of spirochetes internalized by this mechanism with time increased), but was not the preferential mechanism of engulfment by borreliae. Borreliae internalized by coiling phagocytosis were less morphologically altered, and their flagellae protruded into the host cytoplasm. The spirochetes were found discharged in PMN cytoplasm, and were not surrounded by a phagosomal membrane. This could be one of the possible mechanism of persistence of Borrelia burgdorferi in the host organism.
http://www.medscape.com/SLACK/JSTD/1995/v02.n04/jst0204.03.huli/pnt-jst0204.03.huli.html
J Pathol 1994 Jul;173(3):269-82
Related
Articles
A chronic infection with the spirochaete Borrelia burgdorferi typically results in a multistage, multisystem illness. Thus, Lyme borreliosis may provide an interesting model to study the pathomechanisms of microbial persistence. In the present investigation, human peripheral blood monocytes, polymorphonuclear leukocytes, and synovial macrophages were incubated with B. burgdorferi and examined by light and electron microscopy. It was found that incubation with the spirochaetes induced distinct features in the phagocytes. Features which may be related to the pathogenesis of Lyme disease included the segmental uptake of spirochaetes with leaky lysosomes, the invagination of large membrane areas, the extra-lysosomal degradation of internalized B. burgdorferi cells and, finally, the formation of mononuclear syncytial cells and homotypic cell clusters. Features of unknown relevance were the occurrence of two types of cytoplasmic inclusion bodies and exocytic vesicles. These novel findings suggest that reactive alterations of the phagocytes may contribute to the pathogenesis of Lyme borreliosis, which could help to focus future histopathological studies. Moreover, these results may provide new insights into the pathogenesis of other infectious diseases characterized similarly by microbial persistence.
Am J Dermatopathol 1996 Dec;18(6):571-9
Related
Articles, Books
Heterogeneity of Borrelia burgdorferi in the skin.
Aberer E, Kersten A, Klade H, Poitschek C, Jurecka W
Department of Dermatology, University of Vienna, Austria.
The reliability of various in vitro techniques to identify Borrelia burgdorferi infection is still unsatisfactory. Using a high-power resolution videomicroscope and staining with the borrelia genus-specific monoclonal flagellar antibody H9724, we identified borrelial structures in skin biopsies of erythema chronicum migrans (from which borrelia later was cultured), of acrodermatitis chronica atrophicans, and of morphea. In addition to typical borreliae, we noted stained structures of varying shapes identical to borreliae found in a "borrelia-injected skin" model; identical to agar-embedded borreliae; and identical to cultured borreliae following exposure to hyperimmune sera and/or antibiotics. We conclude that the H9724-reactive structures represent various forms of B. burgdorferi rather than staining artifacts. These "atypical" forms of B. burgdorferi may represent in vivo morphologic variants of this bacterium.
PMID: 8989928, UI: 97144094
J Infect Dis 1993 May;167(5):1074-81
Related
Articles
PMID: 8486939, UI: 93253286
J Immunol 1993 Feb 1;150(3):909-15
Related
Articles
The macrophage is a known reservoir for a number of infectious agents, and is therefore a likely candidate site for persistence of Borrelia burgdorferi, the Lyme spirochete. We report that unopsonized B. burgdorferi enter macrophages rapidly, resulting mainly in degradation but occasionally in apparent intracellular persistence. We studied uptake of spirochetes by macrophages by simultaneously labeling infected cells with antibodies to B. burgdorferi and with sequential components of the endocytic pathway, and we examined optical sections (0.5-1.0 micron in thickness) of these cells by confocal fluorescence microscopy at multiple time points after infection. We found that only 5 min of incubation at 37 degrees C were required for nearly 100% of B. burgdorferi to enter a lysosomal glycoprotein-positive compartment, whereas 60 min were required for 90% of the spirochetes to appear in a cathepsin L-positive compartment under the same conditions. We also labeled infected living cells with acridine orange to distinguish live from killed intracellular organisms. Although the large majority of spirochetes within a given cell were dead, we saw occasional live ones up to 24 h (the longest interval examined) after all extracellular organisms had been lysed in distilled water. Moreover, we can reculture spirochetes from macrophages after infection. Persistence of spirochetes within macrophages provides a possible pathogenetic mechanism for chronic or recurrent Lyme disease in man.
PMID: 8423346, UI: 93139523